Research
Faculty-Student Research
Student-faculty collaborative research is one of our programmatic strengths. Including analytical, physical, organic, inorganic and biochemistry projects, our faculty engage students in diverse and interdisciplinary research topics.
Students pursuing the American Chemical Society-approved major complete one year of undergraduate research.
Research Features
Secondary metabolites are molecules produced by organisms that are not essential for survival. However, these molecules provide an evolutionary advantage to the producing organism in a complex biological environment where nutrients are limiting. Organisms produce secondary metabolites as a defense mechanism to promote their survival by inhibiting the growth of or killing competing species. This secret “chemical warfare” is continuously evolving in nature.
Scientific discovery of this microbial “arms-race” has led directly to the treatment of bacterial and fungal infections as well as cancer. These compounds produced in the “chemical warfare” between species in nature have been employed therapeutically as antibiotics and chemotherapeutics. A few of the commonly known antibiotic and chemotherapeutic compounds include doxorubicin, erythromycin, vancomycin, and penicillin. In fact, antibiotic compounds like penicillin were discovered and have been used in treating diseases since the 1940s.
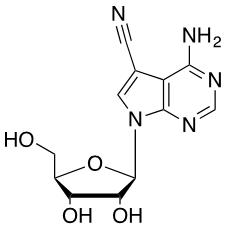
Since the 1970s, discovery of new antibiotic and chemotherapeutic molecules has slowed. Concurrently, there has been a drastic rise in the number of antibiotic–resistant strains of bacteria. To combat these new strains of bacteria, new compounds with enhanced therapeutic activities need to be discovered. One class of compounds, known as the pyrrolopyrimidine containing metabolites (PYPOL), are interesting from the drug discovery perspective. My research focuses on the discovery and characterization of enzymes that catalyze steps in the synthesis of the PYPOL containing compounds in various bacterial organisms. The biosynthesis of the PYPOL containing molecule toyocamycin (TOY, Figure 1) is proposed to require 9 enzymes. However, the function of 5 of the 9 enzymes have yet to be experimentally determined. Current research focuses on functional characterization of the 5 enzymes in order to better understand how bacteria produces TOY. Future investigations will focus on understanding the substrate specificity of these enzymes with a goal of trying to create novel derivatives of TOY. Students that work in my lab will learn a variety of biochemistry and molecular biology techniques including polymerase chain reaction, cloning, Escherichia coli cell culture, protein expression, protein purification, and enzyme kinetics.
My research interests are based on the practical application of fluorescence and luminescence techniques for the detection and quantification of select species in mixtures. This can take on either a theoretical or experimental application. On the theoretical side, our goal is to develop simple, matrix-based algorithms that can model the spectroscopic response from the components of a mixture. The results are compared with experimental results to determine the validity of the model. On the experimental side, we use pulsed (laser based) and non-pulsed (traditional lamp) techniques to generate fluorescence lifetimes (pulsed mode) or fluorescence signatures (lamp) from species on surfaces, species separated via chromatographic techniques, or species intrinsic to a mixture.
Other projects include establishing a Resonance Enhanced Multiphoton Ionization (REMPI) workstation for ultratrace detection of aromatic species in the gas phase. I am also involved in understanding the nature of dative bonding via several techniques including IR grazing angle and possibly Surface Enhanced Raman Spectroscopy (SERS) on self assembled monolayers, gas phase IR in a supersonic slit expansion (in collaboration with the U of MN), and fluorescence signatures of select B-N species in solution phase.
My work requires a “hands-on” aptitude to work with chemical instrumentation and to trouble-shoot experimental methodology. Although programming skills are not necessary, students desiring to develop programming skills are especially welcome. I believe that students ultimately interested in graduate or post- SCSU activities in a physical/analytical laboratory are a good fit for my lab.
One strategy in cancer treatment is the development of drugs that will selectively induce apoptosis in tumor cells. The ability of cancer cells to avoid apoptosis and continue to proliferate is one of the key steps in cancer development. One potential source for new chemotherapeutic agents is natural products. Natural products and synthetic derivatives of natural products make up over 60 percent of all cancer drugs used today.
For example, we are working on the natural product goniothalamin, which comes from the dried stem bark of trees and shrubs from the goniothalamus genus. In preliminary studies, goniothalamin has exhibited low micromolar IC50 values against a variety of different cancer cell lines. These IC50 values illustrate that goniothalamin’s structure could potentially be used as a template for chemotherapeutic drug design. A better understanding of goniothalamin’s mechanism of action might lead to the synthesis of derivatives that demonstrate even more potent cytotoxicity.
The research in my laboratory is directed toward the design and synthesis of novel natural product analogues that will exhibit lower IC50 values against cancer cell lines than the natural product itself. By making hypotheses on the natural products’ mechanism of action, we design analogues by altering the steric and/or electronic properties of the natural product to make it more biologically active. The ultimate goal in this research is to design compounds that are more effective in inducing apoptosis in cancer cells and therefore more potent chemotherapeutic agents.
Physical surface chemistry of metal oxides, with interdisciplinary research with physics and materials science, focusing primarily the composition and speciation of aquatic systems influenced by chemical processes occurring at the solid-solution interface to develop a detailed understanding of the influence of bulk composition and structure, surface orientation, and interaction of water and other surface modifying solutes on the structure and reactivity at the mineral-fluid interface.